Research
Research summary
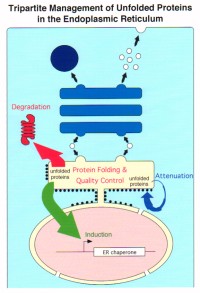 Unfolding or misfolding of proteins constitutes a fundamental threat to all living cells. In eukaryotes, proteins can be unfolded or misfolded in a variety of subcellular compartments such as cytoplasm, mitochondria, and peroxisomes, but the risk of protein misfolding is particularly acute in the endoplasmic reticulum (ER), in which newly synthesized secretory and transmembrane proteins attain their proper tertiary and quaternary structure. Efficient quality control systems have evolved to prevent incompletely folded molecules from moving along the secretory pathway. Thus, accumulation of misfolded proteins in the ER would detrimentally affect the function and/or localization of the approximately one-third of all cellular proteins that translocate into the ER after synthesis on membrane-bound ribosomes. Furthermore, misfolded proteins can exert proteotoxicity through hydrophobic and non-productive interactions with other cellular proteins. Eukaryotic cells have developed ways for dealing with an accumulation of unfolded proteins in the ER: translational attenuation to decrease the burden on the ER, transcriptional induction of ER-localized molecular chaperones and folding enzyme to augment the capacity of productive folding mechanism, and transcriptional induction of components of ER-associated degradation to enhance cellular degradation activity. Our goal is to achieve comprehensive understanding of how eukaryotic cells control the quality of proteins in the ER by pursuing the molecular mechanisms of these homeostatic responses collectively termed unfolded protein response (UPR).
Unfolding or misfolding of proteins constitutes a fundamental threat to all living cells. In eukaryotes, proteins can be unfolded or misfolded in a variety of subcellular compartments such as cytoplasm, mitochondria, and peroxisomes, but the risk of protein misfolding is particularly acute in the endoplasmic reticulum (ER), in which newly synthesized secretory and transmembrane proteins attain their proper tertiary and quaternary structure. Efficient quality control systems have evolved to prevent incompletely folded molecules from moving along the secretory pathway. Thus, accumulation of misfolded proteins in the ER would detrimentally affect the function and/or localization of the approximately one-third of all cellular proteins that translocate into the ER after synthesis on membrane-bound ribosomes. Furthermore, misfolded proteins can exert proteotoxicity through hydrophobic and non-productive interactions with other cellular proteins. Eukaryotic cells have developed ways for dealing with an accumulation of unfolded proteins in the ER: translational attenuation to decrease the burden on the ER, transcriptional induction of ER-localized molecular chaperones and folding enzyme to augment the capacity of productive folding mechanism, and transcriptional induction of components of ER-associated degradation to enhance cellular degradation activity. Our goal is to achieve comprehensive understanding of how eukaryotic cells control the quality of proteins in the ER by pursuing the molecular mechanisms of these homeostatic responses collectively termed unfolded protein response (UPR).
Current Projects
We have identified two intracellular signaling pathways from the ER to the nucleus important for the transcriptional induction in mammalian cells, which ultimately utilize the transcription factors ATF6 and XBP1 activated by regulated intramembrane proteolysis and frame switch splicing, respectively. We are currently investigating the roles of these transcriptional induction systems in the ER quality control.